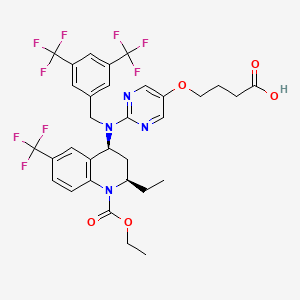
Obicetrapib. Image Credit: PubChem
March 4, 2022 – NewAmsterdam Pharma (NAP), a clinical-stage company focused on the research and development of transformative therapies for cardiometabolic diseases, today announced dose administration for the first patient in its PREVAIL study, a global Phase 3 cardiovascular outcomes trial of the novel cholesteryl ester transfer protein (CETP) inhibitor obicetrapib. PREVAIL is a placebo-controlled, double-blind, randomized study in participants with a history of atherosclerotic cardiovascular disease (ASCVD) who do not have adequate control of their low-density lipoprotein cholesterol (LDL-c) despite being on maximally tolerated lipid-modifying therapies. Today’s announcement follows the recent announcement of NewAmsterdam dosing the first patient in its BROADWAY trial, Phase 3 study of obicetrapib’s LDL-c lowering efficacy.
“The beginning of the Phase 3 PREVAIL cardiovascular outcomes trial is another major step forward in the development of the latest-generation CETP inhibitor,” said Professor Stephen Nicholls of Monash University, Principal Investigator of the PREVAIL trial. “Over 100 million individuals globally are not achieving their LDL-c goals despite currently available lipid lowering therapies. With PREVAIL we aim to demonstrate the impact that obicetrapib’s LDL-c lowering efficacy can provide on patient outcomes in order to help individuals live longer, healthier lives.”
“Starting our cardiovascular outcomes trial in parallel with our Phase 3 BROADWAY lipid lowering study, in addition to further studies we intend to start in 2022, underscores NewAmsterdam’s commitment to demonstrating the potential for obicetrapib — a simple, low-dose, once daily oral therapy — for patients struggling to meet their targets despite their current lipid modifying therapies,” said Michael Davidson, MD, chief executive officer of NewAmsterdam Pharma. “We are thrilled NewAmsterdam will be positioned to share cardiovascular outcomes data just two years after we expect to release our Phase 3 BROADWAY lipid lowering data. We believe these trials will provide a robust package of evidence for obicetrapib’s benefits to patients.”
The primary objective of the PREVAIL trial is to evaluate the effect of obicetrapib on the risk of major adverse cardiovascular events (MACE), including cardiovascular (CV) death, non-fatal myocardial infraction, non-fatal stroke, or non-elective coronary revascularization. Secondary objectives include evaluating the effect of obicetrapib on all-cause mortality, total CV events, new-onset diabetes mellitus (NODM), and change in LDL-c, non-HDL-c, and ApoB levels.
A total of 9,000 patients will be randomized to placebo or 10 mg obicetrapib dosed as a once daily oral treatment. Enrollment is expected to be completed in the first half of 2023.
For more information: www.newamsterdampharma.com/


 January 28, 2026
January 28, 2026 









