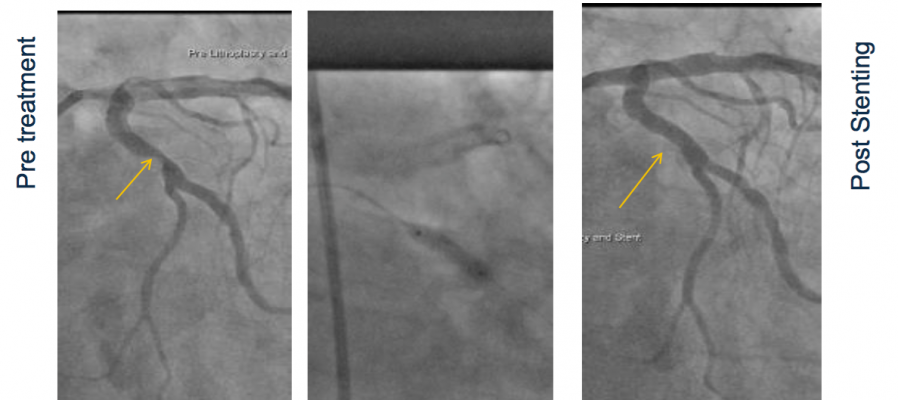
May 23, 2017 — Shockwave Medical announced last week CE mark for the company’s Coronary Lithoplasty System for the treatment of calcified plaque in conjunction with stenting in patients with coronary artery disease.
The Shockwave Medical Coronary Lithoplasty System is designed to treat calcified coronary artery blockages with lithotripsy, sonic pressure waves historically used to treat patients with kidney stones.
Watch the VIDEO "Breaking Up Calcified Lesions Without Vessel Trauma."
The presence of calcified coronary artery disease leads to suboptimal outcomes for all treatment options – medical therapy, interventional treatment and cardiac surgery. For angioplasty with a stent, the presence of calcified lesions is associated with suboptimal lesion expansion, poor stent apposition and complications including dissection, distal embolization, coronary hypoperfusion and procedural failure. Specialty balloons and atherectomy are current adjunctive therapies designed to modify coronary calcium. Their use is limited due to risk of complications, degree of technical difficulty, operator dependency or lack of sufficient evidence.
“Cardiovascular calcification presents a persistent treatment challenge for the interventionalist,” said Jean Fajadet, M.D., co-director of the Interventional Cardiovascular Group at Clinique Pasteur in Toulouse, France, and co-principal investigator of the DISRUPT CAD I clinical trial of the technology. “The use of Lithoplasty in the coronary arteries is an important new option that has shown, in the DISRUPT CAD I clinical study of the device, to effectively prepare the vessel for stent implantation with minimal complications. I look forward to commercial availability of the system.”
Safety and performance was supported by clinical data from results of DISRUPT CAD I, a pre-market, prospective multi-center single-arm study conducted at seven centers in Europe and Australia. The study evaluated the use of the Shockwave Medical Coronary Lithoplasty System as a treatment for calcified coronary arteries prior to drug eluting stent (DES) implantation. Primary endpoint results from the study were reported last fall at the annual Transcatheter Cardiovascular Therapeutics (TCT) conference in Washington, D.C.
The Lithoplasty System integrates angioplasty balloon catheter devices with the calcium-disrupting power of sonic pressure waves, known as lithotripsy. Each Lithoplasty catheter incorporates multiple lithotripsy emitters activated with the touch of a button after the balloon is inflated. Once activated, these emitters produce therapeutic sonic pressure waves that are inherently tissue-selective, passing through the balloon and soft vascular tissue, preferentially disrupting the calcified plaque inside the vessel wall and creating a series of micro-fractures. When the calcium has been modified, the vessel can be dilated using low pressures, thereby enabling even historically challenging patients to be treated effectively with minimal injury to the vessel.
Watch the VIDEO How a Lithoplasty Balloon Shatters Calcified Plaque in Arteries With Ultrasound
Read the article FDA Clears Lithoplasty Balloon That Shatters Calcified Lesions With Ultrasound
For more information: www.shockwavemedical.com


 November 14, 2025
November 14, 2025 









