June 6, 2014 — Continuing to expand its portfolio of endovascular solutions, Medtronic Inc. announced that two recently U.S. Food and Drug Administration (FDA) approved devices will be featured at the Society for Vascular Surgery’s 2014 Vascular Annual Meeting, taking place June 5-7 in Boston. Valiant Captivia Thoracic Stent Graft system proximal FreeFlo tapers line extension now with dissection indication, and the Total across crossing support catheter for peripheral artery disease (PAD) in the below-the-knee (BTK) vessel bed are now available to physicians in the United States.
The Valiant Captivia System proximal FreeFlo tapers line extension is now indicated in the United States for the treatment of thoracic aortic dissections, offering physicians increased patient applicability for fragile tapered, dissected aortas. Thoracic aortic dissection is a condition associated with high morbidity and mortality in which the upper segment of the aorta is torn along the innermost layer of the vessel wall.
“There is a growing body of evidence in support of endovascular repair for thoracic aortic dissection as a minimally invasive alternative to open surgical repair and even medical therapy alone,” said Joseph Bavaria, M.D., professor of surgery and director of the thoracic aortic surgery program at the University of Pennsylvania in Philadelphia, and a national principal investigator for the Medtronic DISSECTION Trial. “The Valiant Captivia system has undoubtedly demonstrated safety and efficacy in the treatment of acute type B dissection cases with excellent technical success.”
In January, Medtronic initially introduced its 11 new proximal FreeFlo tapered pieces to the Valiant Captivia system. The expanded size matrix enables physicians to treat thoracic pathologies in tapered aortas, increasing the configuration possibilities by 30 percent to allow for effective treatment of a wider range of patient anatomies. With the addition of dissection indication for the tapers line extension, Medtronic’s thoracic solutions portfolio is now broadly indicated to treat diseases in the thoracic aorta.
"In aortic dissection, the aorta tapers significantly,” explained Jean Panneton, vascular surgery chief and program director at Eastern Virginia Medical School. “The new Valiant Captivia tapered pieces are ideal for treating these challenging cases, because they offer customizable options. This allows optimal graft sizing that can lead to better procedural outcomes for patients with dissected aortas."
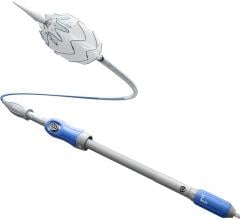
The Total across crossing support catheter has been designed to address specific challenges typically encountered by physicians treating patients with critical limb ischemia (CLI) caused by BTK lesions. It is intended to facilitate access of challenging lesions below the knee by allowing enhanced support for guide wire advancement, as well as guide wire exchange and selective angiography capabilities. It is the only 0.014-inch guidewire compatible crossing support catheter made of a spiral cut stainless steel hypotube that combines exceptional full-length push response with distal flexibility. Moreover the catheter X-ray visualization and its 2 French tapered tip allow easy access and precise positioning in the most difficult distal lesions. Total across has CE mark and recently received FDA 510(k) clearance.
Anvar Babaev, interventional cardiologist at NYU Langone Medical Center, performed one of the first U.S. cases with the Total across crossing support catheter.
“Critical limb ischemia with below the knee chronic occlusion is a particularly challenging subset of patients to treat, and this patient was no exception having occluded, severely calcified distal anterior tibial artery and large non-healing foot ulcer,” said Babaev. “I chose to use the TOTAL across crossing support catheter, which enabled successful crossing and recanalization of the very difficult lesion with pushability and support I had not experienced before.”
The Total across crossing support catheter expands Medtronic’s product portfolio of medical devices used to treat peripheral artery disease (PAD) and is an example of the company’s focus on developing innovative lesion-specific solutions for PAD. Along with the Amphirion Plus and Amphirion Deep PTA Balloon Catheters — the addition of Total across crossing support catheter represents a comprehensive solution portfolio for BTK interventions. It is the first of three new products to address the clinical challenge of BTK CLI that Medtronic plans to introduce around the world over the next two years.
For more information: www.medtronic.com


 April 26, 2023
April 26, 2023