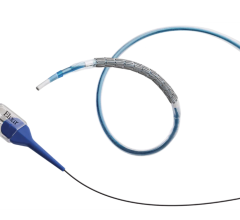
May 28, 2013 — Abbott announced CE mark in Europe for the Xience Xpedition 48 Everolimus Eluting Coronary Stent System, the first-of-its-kind treatment for very long blockages in the vessels that supply blood to the heart due to coronary artery disease (CAD). Xience Xpedition 48 offers the same design and clinical outcomes of the Xience family of drug eluting stents in a 48 mm length. Abbott is the only major manufacturer to offer a coronary drug eluting stent greater than 38 mm in length.
Studies indicate that physicians choose to use multiple shorter length stents to meet the challenges of treating long blockages — or lesions — in as many as 30 percent of interventional heart procedures, as conventional stent sizes may not always fully cover the lesion.[1] It has been shown, however, that the use of a single stent instead of multiple stents may result in several procedural benefits, including the use of fewer devices, less exposure to X-ray during the procedure and reduced procedure time, which could lead to economic benefits.[2]
"With global economic pressures impacting hospitals and health care systems around the world, products like Xience Xpedition 48 may provide important cost savings to the system," said Peter Smits, M.D., Maasstad Ziekenhuis, Rotterdam, the Netherlands. "I believe that this new treatment option, backed by the robust clinical outcomes of the Xience family of drug eluting stents, will help physicians in the treatment of long lesions."
Xience Xpedition 48 is supported by clinical evidence from the Xience family of drug-eluting stents, including data from more than 53,000 patients across more than 100 studies with long-term outcomes out to five years.
Xience Xpedition is available in one of the broadest size matrices on the European market, with diameters ranging from 2.25-4 mm, including a 3.25 mm diameter, and now with lengths ranging from 8-48 mm.
In the United States, the Xience Xpedition and Xience Prime stent systems, including Xience Prime LL, are indicated for improving coronary artery luminal diameter in patients with symptomatic heart disease due to de novo native coronary artery lesions (lesions ? 32 mm) with reference vessel diameters of ? 2.25 to ? 4.25 mm.
For more information: www.xiencestent.com
References:
1. Räber, L., Jüni, P. et al. "Impact of stent overlap on angiographic and long-term clinical outcome in patients undergoing drug-eluting stent implantation." Journal of the American College of Cardiology 2010; 55(12):1178-88
2. Hoffman, R., Herrmann, G. et al. "Randomized comparison of success and adverse event rates and cost effectiveness of one long versus two short stents for treatment of long coronary narrowings." The American Journal of Cardiology 2002; 90:460-464
3. Morgan, K.P., Kapur, A. & Beatt, K.J. "Anatomy of coronary disease in diabetic patients: an explanation for poorer outcomes after percutaneous coronary intervention and potential target for intervention." Heart 2004; 90:732-738.


 July 02, 2024
July 02, 2024