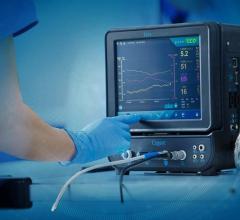
October 11, 2012 — Edwards Lifesciences Corp. announced it has completed the acquisition of BMEYE B.V., a privately held Dutch company that specializes in the development of noninvasive technology for advanced hemodynamic monitoring. BMEYE's technology provides continuous, real-time information to clinicians and can be used in the surgical, intensive care, emergency room and cardiology settings.
"BMEYE's unique noninvasive technology platform complements our existing portfolio and will provide clinicians with critical, comprehensive hemodynamic monitoring information for a broader range of patients. Numerous studies have concluded that therapy guided by hemodynamic monitoring improves patient outcomes and reduces complications and lengths of hospital stays," said Carlyn Solomon, Edwards' corporate vice president, critical care and vascular. "This acquisition supports our ongoing commitment to build and strengthen our offerings for clinicians and patients, and reinforces our leadership in hemodynamic monitoring."
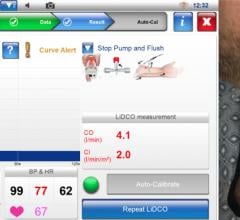
BMEYE's monitoring products work with a noninvasive finger cuff to measure continuous, real-time information on cardiac output, blood pressure, fluid responsiveness and other important hemodynamic parameters. Currently, BMEYE's products are approved for sale in the United States, throughout Europe and in several other countries.
BMEYE CEO Rob de Ree said, "BMEYE has a solid history of developing innovative hemodynamic monitoring solutions. We look forward to joining the Edwards team to extend the reach of our existing products, and to continue to develop novel technologies to help clinicians and improve patient care worldwide."
While BMEYE's products have primarily been used by clinicians outside of the hospital, over the next 18 months Edwards plans to further develop the technology and integrate it into the Edwards EV1000 clinical platform for use in the acute care setting in hospitals.
For more information: www.edwards.com


 January 13, 2026
January 13, 2026