July 9, 2012 — Endosystems Inc., a medical device company developing technology for endovascular aneurysm repair (EVAR), announced closing on $25 million of equity financing. Synergy Life Science Partners, a new investor, led the round and was joined by existing investors US Venture Partners and Longitude Capital Management. Mudit K. Jain, Ph.D., partner at Synergy Life Science Partners, will join the company’s board of directors.
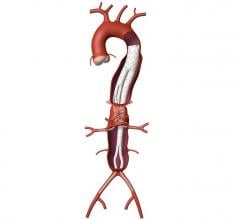
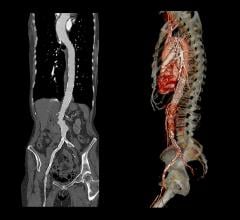
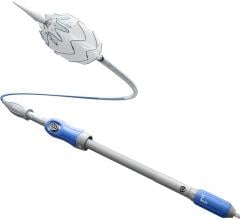
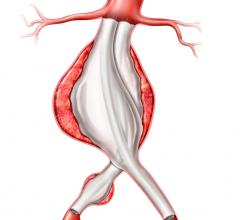
Proceeds from the financing will be used to expand commercialization of the HeliFX Aortic Securement System in the United States and Europe, where the company has already obtained U.S. Food and Drug Administration (FDA) clearance and CE mark, respectively. In addition, Aptus will develop further HeliFX product offerings to fulfill unmet needs in the treatment of aortic aneurysms. The company also plans to execute a U.S. clinical trial of the Fortevo AAA Endograft System in support of filing for premarket approval with the FDA. Lastly, Aptus will expand utilization of the Fortevo system in key European centers, where it has already received the CE mark.
“This commitment on the part of Synergy Life Sciences Partners and our current investors provides the company with necessary capital required to bring the HeliFX and Fortevo family of products to physicians treating patients with life-threatening aortic aneurysms,” said James Reinstein, president and CEO of Aptus Endosystems. “We will judiciously utilize these funds to expand the use of HeliFX in targeted markets and introduce new aortic repair products, all while approaching profitability.”
“Aptus’ HeliFX and Fortevo technology platforms provide an innovative treatment option for millions of patients suffering from AAA,” said Jain. “We believe that Aptus’ proprietary technology and world-class management team uniquely position the company for significant growth in years ahead.”
For more information: www.aptusendosystems.com


 April 26, 2023
April 26, 2023