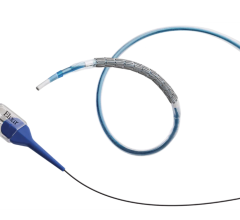
June 11, 2010 – A new drug-eluting stent (DES) was launched this week in the European Union and other CE mark countries. The Taxus Element Paclitaxel-Eluting Coronary Stent System is Boston Scientific’s third-generation DES technology and incorporates a platinum chromium alloy with a new stent design and catheter delivery system.
It received CE mark approval last month, which included a specific indication for the treatment of diabetic patients.
The first European implants were performed by Corrado Tamburino, M.D., Ph.D., chair of the cardiology department at Ferrarotto Hospital, Catania, Italy and Adrian Banning, M.D., directorate chair of cardiac services at John Radcliffe Hospital, Oxford, United Kingdom.
“I have found the Taxus Element Stent to offer performance advantages in flexibility, visibility and deliverability over prior-generation stents,” Tamburino said. “The platinum chromium alloy and new stent design used in the Element platform, together with the proven Taxus drug and polymer, represent a significant advance in coronary stenting.”
“The diabetic indication for the Taxus Element Stent System provides an important benefit given that approximately one third of all patients presenting with coronary artery disease in Europe have diabetes,” Banning said. “Diabetic patients with coronary artery disease often have poorer outcomes after revascularization procedures. The paclitaxel-based Taxus Element Stent has a unique mechanism of action that helps inhibit restenosis in high-risk patients with diabetes.”
The Taxus Element is designed specifically for coronary stenting and leverages the performance advantages of the Element stent platform with a decade of clinical success from the Taxus program. The novel stent architecture and proprietary platinum chromium alloy combine to offer greater radial strength and flexibility. The stent architecture helps create consistent lesion coverage and drug distribution while improving deliverability, which is enhanced by an advanced catheter delivery system. The higher density alloy provides superior visibility and reduced recoil while permitting thinner struts compared to prior-generation stents.
Trial Results
In March, the company announced 12-month results from its PERSEUS clinical program demonstrating positive safety and efficacy outcomes in workhorse lesions for the Taxus Element compared to the Taxus Express2 stent. Those results were recently published in the Journal of the American College of Cardiology. The PERSEUS results also reported a similar safety profile and statistically superior efficacy outcomes in small vessels for the Taxus Element compared to a historical control group of patients receiving the Express bare-metal Stent.
Regulatory Approval
The company received CE mark approval for the Promus Element Everolimus-Eluting Stent System in October 2009. Both Element stent systems incorporate the same platinum chromium alloy, innovative stent design and advanced catheter delivery system.
In the United States, the company expects U.S. Food and Drug Administration (FDA) approval for the Taxus Element in mid-2011 and for the Promus Element in mid-2012. In Japan, the company expects approval for the Taxus Element in late 2011 or early 2012, and for the Promus Element in mid-2012.
In the United States and Japan, the Taxus Element Stent and the PROMUS Element Stent are investigational devices and are limited by applicable law to investigational use only and are not available for sale.
For more information: www.bostonscientific.com


 July 02, 2024
July 02, 2024