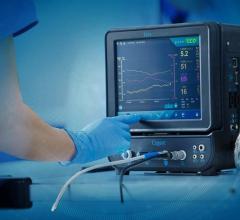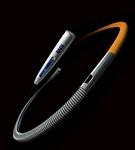
February 23, 2010 – Hospitals using the McKesson Horizon Cardiology Hemo solution will now be able to use St. Jude Medical’s wireless PressureWire Aeris fractional flow reserve (FFR) measurements.
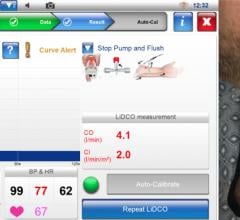
Through an agreement with McKesson, physicians will have greater access to the market’s only wireless FFR measurement system. FFR measurement indicates the severity of blood flow blockages in the coronary arteries, allowing physicians to better identify which specific lesion or lesions are responsible for a patient’s ischemia, a deficiency of blood supply to the heart caused by blood restriction. The PressureWire Aeris technology will help physicians determine whether a stent is needed based on the blood flow around a lesion.
The PressureWire Aeris wireless technology works by integrating physiological lesion assessment directly into the hemodynamic recording system already available in the cath lab to immediately display, measure and save FFR data. This integration is an optional module for hospitals using McKesson’s Horizon Cardiology Hemo system version 12.1. The McKesson Horizon Cardiology Hemo system was designed to improve the overall workflow of the entire catheterization suite. A single interface in the Horizon Cardiology Hemo system combines both the FFR measurement controls and clinical documentation including lesion, stage and medication.
For more information: www.sjm.com


 January 13, 2026
January 13, 2026