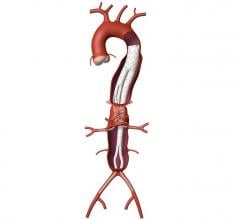
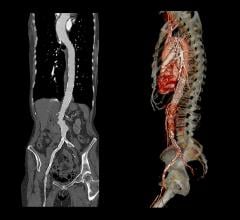
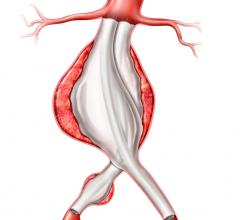
October 9, 2009 – Cook Inc. has filed a lawsuit with the U.S. District Court, Southern District of Indiana, against Endologix Inc., claiming infringement of two critical patents for endovascular stent-grafts used for endovascular repair of aortic aneurysms (EVAR).
“Cook has clearly established itself as the world leader in developing advanced endovascular technologies to treat aortic aneurysms and dissections, and this suit seeks to protect that position from damages caused by Endologix’s infringement of U.S. Patents No. 5,755,777 and No. 5,035,706,” said Cook general counsel Cynthia Kretz. “As part of our vigorous defense of our patent portfolio in this area, Cook has asked the U.S. District Court to enjoin Endologix from selling all products infringing on these patents and to award Cook damages adequate to compensate for the infringement, including interest and costs.”
The suit was filed Oct. 6 and asks that a jury trial be held to determine the outcome of the complaint. The two patents in question are critical for Cook’s leadership in the EVAR field.
The ‘777 patent protects Cook’s technology for its expandable transluminal graft prosthesis for repair of aneurysms, which was awarded in May 1998. The ‘706 patent covers Cook’s percutaneous stent and method of retrieval thereof, which was granted in July 1991.
For more information: www.cookmedical.com


 April 26, 2023
April 26, 2023