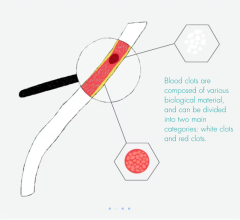
July 8, 2009 - Pluristem Therapeutics Inc. said yesterday its first patient enrolled in a phase I clinical trial that will evaluate the safety of the company’s allogeneic, placental-derived, adherent stromal cell product, termed PLX-PAD. The product is for use in patients afflicted with critical limb ischemia (CLI), the end-stage of peripheral artery disease (PAD). Up to 15 adults with the disease will be included in the trial, which is being conducted at the Franziskus-Krankenhaus Hospital and Charité - Universitätsmedizin Hospital in Berlin.
The Paul Ehrlich Institute recently approved the company to begin clinical trials with PLX-PAD.
PLX-PAD is a cellular therapeutic product derived from the placenta and is a ubiquitous source of adult stromal cells easily obtained without harm to the mother or baby.
For more information: www.pluristem.com


 September 12, 2025
September 12, 2025