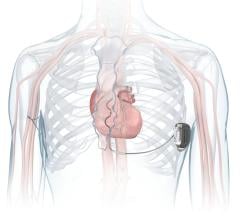
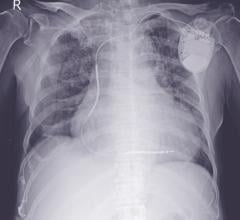
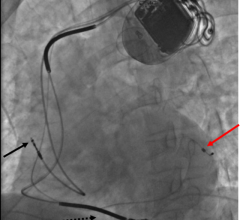
April 14, 2009 - Biotronik Inc. announced yesterday the first U.S. implant of the next-generation Lumax 540 Series defibrillator for treating patients with heart failure.
The Lumax 540 Series was recently approved by the FDA.
The first U.S. implant of the new Lumax 540 Series was performed by Leon Feldman, M.D., FACC, co-director of electrophysiology and cardiology section chief at Eisenhower Medical Center in Rancho Mirage, Calif.
“The patient presented with very complex needs: ongoing bradycardia, recurrent ventricular tachycardia, and progressive heart failure all at once,” Dr. Feldman said. “The patient will benefit from the advanced capabilities of the Lumax 5 Series defibrillator.”
Heart failure is on the rise in the U.S., affecting 5 million people with 550,000 new cases each year. Fortunately, therapies are available to enhance the quality of life of a heart failure patient.
“The patient already received the highest dose of beta blockers and ACE inhibitors that he could handle and was still in severe heart failure,” Dr. Feldman said, who said a cardiac resynchronization therapy defibrillator (CRT-D) was the perfect solution. “I’ve had great success with previous-generation Biotronik devices and have confidence in the construction of the device and lead technology. Also, I’m a big proponent of Home Monitoring for the patient, which can remotely and rapidly detect onset of atrial fibrillation, for which this patient is at risk.”
Dr. Feldman said the benefits that Biotronik's Home Monitoring system offers him and the patient early notification of VF and VT, which allows him to modify therapy and avoid unnecessary shocks.
For more information: www.biotronik.com


 January 13, 2026
January 13, 2026