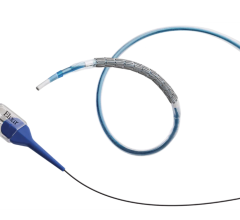
September 25, 2008 - Boston Scientific Corp. announced it has received approval from the FDA to market its TAXUS Express2 Atom paclitaxel-eluting coronary stent system, a drug-eluting stent (DES) specifically designed for treating small coronary vessels.
The TAXUS Express2 Atom Stent System is the only DES approved by the FDA for use in vessels as small as 2.25 mm in diameter, stated Boston Scientific.
The company today also announced FDA clearance of its TAXUS Express2 paclitaxel-eluting coronary stent system for the treatment of in-stent restenosis in bare-metal stents. It is reportedly the only drug-eluting stent approved in the U.S. for the treatment of in-stent restenosis in bare-metal stents.
"The TAXUS Express Atom Stent will provide better options for U.S. patients with coronary artery disease in small vessels," said Gregg Stone, M.D., chairman of the Cardiovascular Research Foundation and professor of medicine at Columbia University Medical Center, and principal investigator of the TAXUS IV and V clinical trials. "This is a welcome addition to the range of available drug-eluting stents, since patients with small vessels who are currently treated with bare-metal stents experience high rates of restenosis. In the TAXUS V clinical trial, the TAXUS Express Atom Stent significantly reduced the chance of restenosis and the need for repeat procedures compared to bare-metal stents, in patients with small vessel disease."
For more information: www.bostonscientific.com.


 July 02, 2024
July 02, 2024