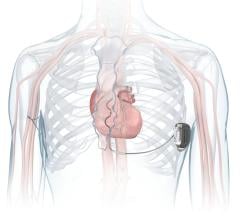
February 5, 2008 - Doctors completed the first implant of Boston Scientific’s COGNIS cardiac resynchronization therapy defibrillator (CRT-D), which reportedly represents an entirely new platform to treat heart failure.
The COGNIS was part of a multi-year research and development effort to build a devices that offers extended longevity and a number of new features to manage heart failure patients. One such feature is the SmartDelay, which is said to quickly proposes programmable device settings and enable clinicians to tailor individualized pacing therapy for their patients.
The COGNIS CRT-D is small and thinn with high-energy at 32.5 cc, and less than 10 mm thick. The COGNIS CRT-D also offers SafetyCore, a feature that in the unlikely event of a system error is designed to provide lifesaving shock therapy and basic pacing functionality.
Key features of the COGNIS CRT-D include:
- Bi-V Trigger: designed to help physicians manage heart failure patients with frequent atrial arrhythmias
- Electronic Repositioning: provides physicians with the ability to electronically select between six different configurations for stimulating the left side of the heart even after implant, which could help avoid an additional surgical procedure, the company says.
- Left Ventricle only pacing: this feature provides physicians with an additional pacing option for CRT patients
For more information: www.bostonscientific.com


 January 13, 2026
January 13, 2026