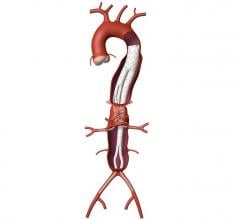
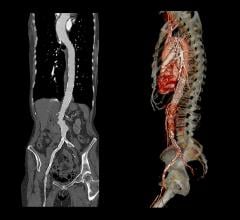
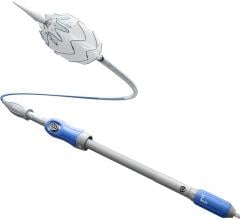
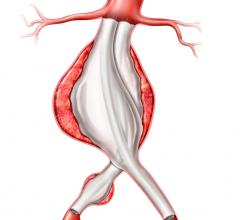
January 14, 2008 - Medtronic Inc. launched the AneuRx AAAdvantage Stent Graft on the new Xcelerant Hydro Delivery System, expanding its product line in endovascular therapies for aortic aneurysms.
The Xcelerant Hydro Delivery System features a hydrophilic coating designed to aid navigation of the device through tight and tortuous arteries by reducing friction with the artery wall. The system will be used with the AneuRx AAAdvantage Stent Graft, which was introduced in the United States in March 2006. In bench testing, the Xcelerant Hydro Delivery System was shown to generate 68 times less friction than the previous delivery system, which does not have the hydrophilic coating.
In addition, the Xcelerant Hydro Delivery System features an integrated sheath that is tapered on both ends. This dual-taper sheath is designed to facilitate insertion and retraction of the entire delivery catheter by minimizing the time that the surface area of the sheath is in contact with the artery wall. The integrated sheath also contributes to the system’s low profile characteristics, which are intended to optimize tracking and access through small vessels.
“It will simplify the procedure for endovascular interventionalists in treating patients whose iliac arteries are difficult to navigate when they are small and tortuous,” said Frank R. Arko, M.D., chief of Endovascular Surgery at the University of Texas Southwestern Medical Center in Dallas.
For more information: www.medtronic.com


 April 26, 2023
April 26, 2023