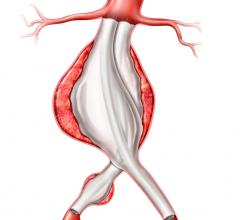
January 3, 2008 - A modular stent graft that inserts multiple, caudally-directed branches to the visceral arteries to repair a thoracoabdominal aortic aneurysm (TAAA) can offer a safer surgical method for patients in need of this surgery, according to a recent study in the January 2008 issue of the Journal of Vascular Surgery.
University of California at San Francisco (UCSF) researchers (who have studied stent grafts for TAAA since 2000), maintain that this less invasive approach eliminates aneurysm flow, while preserving visceral perfusion and avoiding the main physiologic stresses. In the endovascular approach there is no need to open the abdomen, expose the aneurysm, or clamp the aorta, which should translate into less mortality, morbidity, pain and debility.
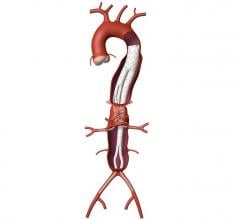
"Our team has used various stent graft components connected in a number of ways," said Timothy A. M. Chuter, M.D., division of vascular surgery at UCSF, and research team leader. "Our current technique employs a springy (self-expanding) stent-graft that curves downwards and outwards from the main trunk to mimic the branching pattern of a normal thoracoabdominal aorta."
The current study between January 2006 and May 2007, showed that 22 patients with an average age of 76 years (all unfit for open repair and nine had undergone prior aortic surgery), were successfully treated with self-expanding covered stents connecting the caudally-directed cuffs of an aortic stent graft with the visceral branches of an aortic aneurysm (81 branches in all). These customized aortic stent grafts were inserted through the femoral (16) or iliac (6) arteries, where covered stents were inserted through the brachial arteries. Two patients died perioperatively: one from guidewire injury to a renal arterial branch and the other from a medication error.
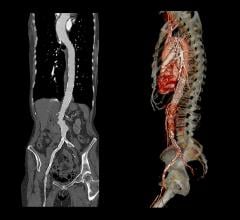
Routine follow-up included contrast enhanced CT scans at one week, one month, six months and one year. One branch (renal artery) occluded at one month. The other 80 branches remain patent. There were no signs of stent graft migration, component separation or fracture.
Serious or potentially serious complications occurred in 9 of 22 patients, but there was no paraplegia, renal failure, stroke or myocardial infarction among the 20 surviving patients. Complications included one patient who had a localized intimal disruption, who underwent successful reintervention. Another patient had aortic dissection (early), type I endoleak (early) and stenosis of the superior mesenteric artery (late). Also, one patient has a type II endoleak.
Dr. Chuter added that the rate of serious complications in this recent series was relatively low, and endovascular repair is feasible in most TAAAs. The best candidates for endovascular repair are patients with an extensive TAAA (type II or type III), previous aortic surgery and an otherwise normal life expectancy."
For more information: www.jvascsurg.org


 April 26, 2023
April 26, 2023