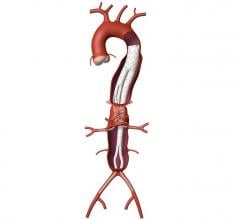
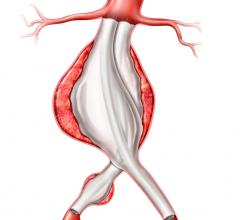
April 16, 2007 - Endologix, Inc. has announced the first three successful implantations of the Powerlink endoluminal stent graft (ELG) incorporating the Visiflex SurePass Delivery System.
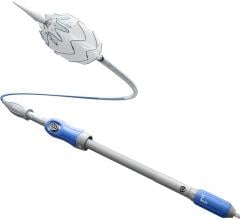
The Visiflex SurePass is the company's next-generation delivery device that allows physicians continued guide wire access to the contralateral limb of the unibody Powerlink ELG device.
The first procedure was completed at William Beaumont Hospital in Royal Oak, MI., and two additional procedures were completed at the Arizona Heart Hospital in Phoenix, AZ.
* Venkatesh G. Ramaiah, M.D., director of vascular and endovascular research at the Arizona Heart Institute, reported, "The SurePass Delivery System further improves upon a delivery technology that is already state-of-the-art in terms of ease of deployment."
* Grayson Wheatley III, M.D., from the Arizona Heart Institute, remarked, "It exceeded my expectations and in this particular case facilitated access to a difficult contralateral limb in a tortuous anatomy."
* O. W. Brown, M.D., from the William Beaumont Hospital, commented, "The technology performed beyond my expectations and I believe the SurePass will further simplify and broaden the applications for Powerlink."
The Visiflex SurePass Delivery System replaces the contralateral limb wire with a hollow guidewire capable of accommodating a standard 0.014 guidewire. The physician can insert the guidewire prior to limb deployment in order to control and maintain access. In patients with complex iliac anatomy physicians often perform adjunctive balloon angioplasty or deliver an extension, which requires the use of a guidewire.
For more information visit www.endologix.com.


 April 26, 2023
April 26, 2023