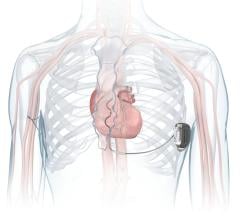
Jan. 15, 2007 — Medtronic, Inc. announced today its launch of a national campaign designed to raise awareness of sudden cardiac arrest (SCA), its risk factors, and the role of implantable cardioverter-defibrillator (ICD) therapy in saving lives. SCA is the one of the nation's leading causes of death -- claiming more lives than breast cancer, lung cancer, and HIV/AIDS combined.
"Today, nearly 1,000 Americans will die of sudden cardiac arrest — many of these deaths could have been prevented with ICD therapy," said Steve Mahle, president of Cardiac Rhythm Disease Management at Medtronic. "We believe that more can be done to reach the hundreds of thousands of people who we know could benefit from ICD therapy, but remain unprotected. Ultimately, we believe this campaign will help save more lives."
The comprehensive campaign includes advertising; patient and caregiver information resources; physician education programs and outreach; and ongoing clinical research on SCA and the use of ICD therapy. The advertising begins this week with the launch of print, television and online advertising in the U.S.
The ads, themed "What's Inside," show the link between medical device technology and the benefits people can experience by living with ICD therapy. The ads encourage those who may be at greatest risk of SCA — people who have had a heart attack or been diagnosed with heart failure — to visit www.whatsinside.com or call 866-950-5550 for more information, including an SCA risk assessment that can be used to facilitate a discussion with a physician.
Sudden cardiac arrest is a sudden, abrupt loss of heart function — it is 95 percent fatal if left untreated, the company stated. Most SCA episodes are caused by the rapid and/or chaotic activity of the heart known as ventricular tachycardia (VT) or ventricular fibrillation (VF). Recent estimates show that approximately 850,000 people in the U.S. are at risk of SCA and indicated for an ICD device, but remain unprotected. In fact, less than 35 percent of patients who are indicated for an ICD have one.


 January 13, 2026
January 13, 2026