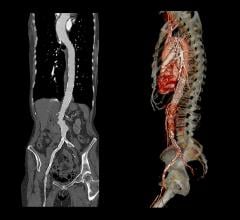
November 24, 2010 - Rotational computed tomography angiography (CTA), a new noninvasive imaging modality, has the ability to display angiography in 3-D formats. This allows for considerable simplification of orientation of complex fenestrated/branched stent grafts in 3-D views, as well as catheterization of target vessels. It also enables practitioners to determine whether fenestrated/branched stent grafts are successfully deployed without leaks and thus obviates the need for immediate post-operative CTA.
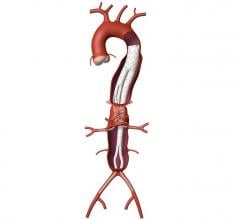
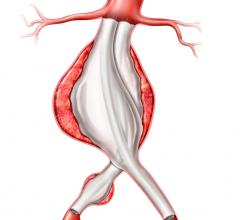
The plans, constructed using multidetector CTA (MCTA) thin slice images, provide guidance for the final positioning of the fenestrated/branched stent grafts in the aorta. However, especially when the aorta is tortuous, the planned position is often an approximation to the final position achieved. This can considerably increase the technical challenges associated with placement of connecting stents.
Until recently, only 2-D angiographic images have been possible to guide placement of the endograft and connecting stents. In order to overcome this limitation, multiple views from different angles such as from the frontal anterior-posterior to lateral views, have been employed. A large amount of nephrotoxic contrast medium is often used for these repeated 2-D angiographies. Recent developments in angiographic equipment have made possible the acquisition of rotational angiography that gives a 180-degree view of the vascular tree.
“Using this new rotational CTA has proved to be very helpful in determining orientation of fenestrated/branched stent grafts,” said Krassi Ivancev, M.D., professor at the University College of London. “One way to facilitate such orientation in 3-D is to catheterize one or two of the target vessels and then, by using the capabilities of a rotational CTA without contrast medium, making it possible to orient the fenestrated/branched stent grafts accurately using the catheters in the target vessels as reference points.”
“The same technique can be employed for catheterization of target vessels whereupon a limited amount of contrast medium is used selectively only through the catheters in the target vessels, thus reducing the large amount of contrast medium otherwise necessary for routine 2-D angiography/aortogram from differently angled views,” he said.
Ivancev said it is also possible to fuse images from a pre-op MCTA with the images from a rotational CTA, and then to use the fused images for catheterization of the target vessels. In the near future rotational CTA may facilitate selection of an appropriate off-the-shelf fenestrated/branched stent graft based upon 3-D evaluation of the aorta with a large dummy sheath in place to simulate the relative behavior of stent grafts and the target vessels in the presence of tortuosity.
For more information: www.veithpress.org


 April 26, 2023
April 26, 2023