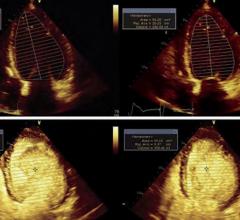
December 16, 2008 - New data from a retrospective, multicenter, cohort study of over four million hospitalized patients shows similar unadjusted mortality rates for patients receiving DEFINITY Vial For (Perflutren Lipid Microsphere) Injectable Suspension enhanced resting echocardiography exams when compared with patients who did not receive DEFINITY.
Study findings were published in the December 2008 issue of The American Journal of Cardiology.
Although the patients who received DEFINITY were sicker on average and had a higher baseline mortality risk prior to the exam, following DEFINITY administration these same patients exhibited a 24 percent reduced risk of mortality over 24 hours compared to the those patients who received only echocardiography without the ultrasound contrast agent.
"As the largest retrospective, observational study to date to examine the short-term safety profile of perflutren-containing ultrasound contrast agents, the data highlight the relatively low incidence of adverse safety reactions associated with DEFINITY and demonstrate a significantly lower risk of acute mortality for patients receiving DEFINITY-enhanced echocardiograms," said Mark Hibberd, M.D., Ph.D., Senior Medical Director, Global Medical Affairs, Lantheus Medical Imaging, Inc. and co-author on the study. "These study findings provide the physician community with essential safety information on the use of DEFINITY, especially among hospitalized patients, and provide valuable information on patient outcomes that bear further investigation."
The study, which was designed to assess short-term (one-day) mortality in hospitalized patients undergoing resting transthoracic echocardiography both with and without DEFINITY, analyzed more than 4.3 million patients undergoing clinically indicated echocardiography from Jan. 1, 2002 through Oct. 31, 2007. The study population was from the Premier Perspective Database, the largest U.S. hospital-based, service-level comparative database providing detailed resource utilization data along with patients' primary and secondary diagnosis and procedure codes. Of this population, 4,242,712 patients received non-enhanced echocardiograms and 58,254 received contrast-enhanced studies using DEFINITY.
One-day mortality rates were 1.08% for patients undergoing non-contrast studies and 1.06% for patients undergoing DEFINITY contrast-enhanced examinations. However, patients receiving DEFINITY were more likely to have an increased baseline severity of illness and a higher pre-study risk of mortality. A multivariate logistic regression analysis was used to compare 24-hour mortality, controlling for case mix and clinical covariates. Patients undergoing DEFINITY-enhanced echocardiograms had a 24% lower risk adjusted odds ratio for mortality within one day than those patients who received only echocardiography without the ultrasound contrast agent.
"This study is groundbreaking in the echocardiography area, and represents the largest evaluation of resting echocardiography among hospitalized patients," said Dr. Hibberd. "It is also the first to evaluate the impact of contrast echocardiography on survival among hospitalized patients. These data provide the medical community with further clinical evidence of how DEFINITY can significantly improve the standard of care for patients undergoing cardiac evaluations, and we are continuing to explore the role of DEFINITY in specialized patient populations such as those in the ICU and receiving mechanical ventilation."
For more information: www. www.lantheus.com


 August 17, 2023
August 17, 2023