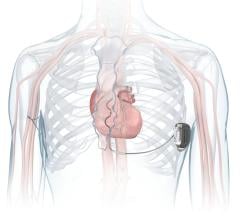
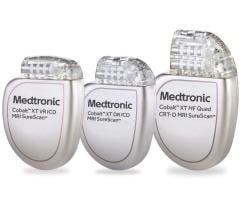
July 9, 2007 - Medtronic Inc. announced the commercial launch of its latest cardiac resynchronization therapy-defibrillator, the Concerto CRT-D, and its latest implantable cardioverter-defibrillator, the Virtuoso ICD, which are the first of their kind in the Japanese market.
The CRT-D devices are equipped with the functions of an ICD, while also providing cardiac resychronization therapy for patients suffering from heart failure. The Concerto and Virtuoso are the first devices in Japan with wireless functionality, designed to reduce follow-up and procedure times.
The implanted devices internally record the patient’s pulse, device therapies delivered and other data over a certain period of time. This information is then provide this information wirelessly. Device settings can also be performed using the wireless function.
The Concerto and Virtuoso deliver anti-tachycardia pacing (ATP), while simultaneously charging the capacitor to provide a shock, if needed. This means that if the arrhythmia is not terminated using ATP, the devices are able to deliver the electrical shock with no delay, giving patients access to the safe, painless ATP therapy while minimizing patient burden.
The Concerto and Virtuoso are also reportedly the first devices in Japan capable of measuring the impedance in the thoracic cavity between the device implanted below the clavicle and the lead implanted in the ventricle, allowing the device to record the level of fluid in the patient’s lungs.
For more information: www.medtronic.com


 January 13, 2026
January 13, 2026