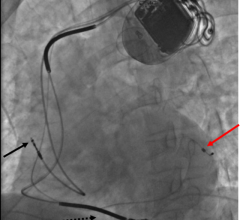
December 11, 2008 - The Heart Rhythm Society is reiterating its position on the importance of device and lead reliability and safety following the publication of “Learning from Our Mistakes? Testing New ICD Technology” by Robert Hauser, M.D., and Adrian Almquist, M.D., as well as a corresponding letter from Mitchell Shein and Daniel Schultz from the FDA, which were published in the Dec. 11 issue of the New England Journal of Medicine.
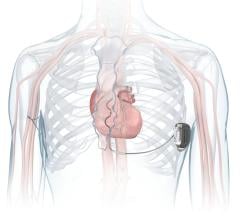
“The Heart Rhythm Society is committed to the highest standard of patient safety,” said N.A. Mark Estes, president of the Heart Rhythm Society. “We have been at the forefront of establishing policies and guidelines to ensure the safety of life-saving devices such as internal cardiac defibrillators (ICDs) and pacemakers. The society will continue to develop standards as technology continues to evolve.”
To address issues regarding the safety of device leads, the Heart Rhythm Society created a task force earlier this year with leading experts and is currently collaborating with regulatory agencies, industry and patients to develop recommendations regarding lead performance. The Heart Rhythm Society anticipates releasing the new recommendations at Heart Rhythm 2009, the society’s annual scientific sessions which will take place in Boston in May.
The society’s recommendations for the surveillance, analysis and performance reporting of pacemakers and implantable cardioverter defibrillators (ICDs), released in September 2006, have been adopted by physicians, government and industry and helped to achieve greater transparency in the post-market surveillance, analysis and reporting of information; the establishment of new systems to identify malfunctioning devices more quickly; and standard notification and communication to physicians and patients from the manufacturer when a device malfunction is identified.
For more information: www.HRSonline.org


 January 13, 2026
January 13, 2026