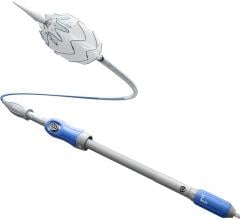
July 15, 2010 -- W. L. Gore and Associates recently announced that it has received the European CE mark approval for the 25 cm Gore Viabahn Endoprosthesis with Propaten Bioactive Surface. The Gore device is the longest length stent-graft available, designed to cover more of the lesion in the superficial femoral artery (SFA) potentially reducing the need for multiple devices.
The recently redesigned Gore device features a precision laser-trimming technology used to remove excess material, resulting in a contoured proximal edge that may improve flow dynamics at the proximal end. Removal of excess material at the proximal edge improves device apposition to the vessel wall when oversizing prevents device expansion to its nominal diameter.
The device also incorporates the Propaten Bioactive Surface, which utilizes end-point immobilization of derivatized heparin to the endoprosthesis luminal surface. This proprietary surface technology preserves the heparin bioactive sites such that they remain free to interact with the blood at the device surface without being consumed. The original Gore Hemobahn Endoprosthesis was introduced to Europe in 1996; the Gore Viabahn Endoprosthesis with Propaten Bioactive Surface was first approved for use in the European Union in December 2008.
The Gore Viabahn Endoprosthesis with Propaten Bioactive Surface is available with a low-profile delivery system that gives interventionalists a more streamlined approach to re-line the peripheral arteries. The Gore Viabahn Endoprosthesis is constructed with a durable, reinforced, biocompatible, ePTFE liner attached to an external nitinol stent structure. The excellent flexibility of the Gore Viabahn Endoprosthesis enables it to better traverse tortuous areas of the SFA and conform more closely to the complex anatomy of the artery.


 April 26, 2023
April 26, 2023