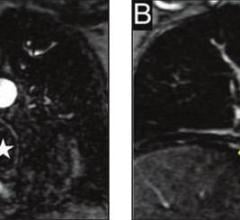
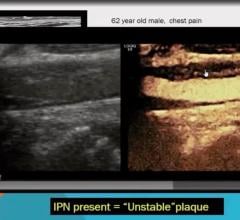
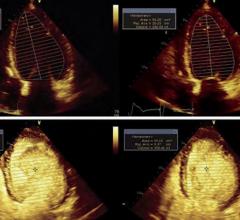
January 22, 2008 - Avista Capital Partners, which recently acquired the Bristol-Myers Squibb Medical Imaging medical imaging business unit, reported today that the FDA has granted pediatric exclusivity for studies conducted on Cardiolite, effective Jan. 8, 2008, extending the marketing exclusivity of Cardiolite for an additional six months beyond patent expiration on Jan. 29, 2008.
Cardiolite is a kit for the preparation of Technetium Tc99m Sestamibi for Injection.
“We believe that this is the first pediatric exclusivity grant for an imaging agent and is the result of extensive clinical trials, involving more than six hundred (600) patients in nine countries over three years,” said D. Scott Edwards, V.P. of Research and Development at MI.
For more information: www.avistacap.com


 August 17, 2023
August 17, 2023