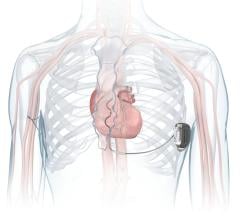
September 18, 2007 - St. Jude Medical Inc. announced FDA approval of its first radiofrequency (RF) wireless devices to treat patients with heart failure and with potentially lethal heart arrhythmias. The Promote RF CRT-D (cardiac resynchronization therapy defibrillator) and Current RF ICD (implantable cardioverter defibrillator) feature radiofrequency telemetry for wandless communication with programmers used by physicians to interrogate and program devices.
RF telemetry is designed to enable secure, remote communication between the implanted device and the programmers in a clinician's office or hospital. Wireless communication occurs while the device is being implanted and when patients see physicians for follow-up visits, allowing for efficient, more convenient care and device management.
The devices use a dedicated range of frequencies designated for medical devices called the MICS (Medical Implant Communications Service) frequency band, which helps to prevent interference from other electronic signals. The MICS technology also includes the capability to choose between 10 channels in order to optimize the telemetry link and avoid interference.
The Promote RF CRT-D and Current RF ICD are built on St. Jude Medical’s next generation Unity device platform. This consolidated electronics platform is engineered to enable St. Jude Medical to more quickly introduce devices with new features and diagnostics, as they become available, because the basic platform for all of the devices is the same. In addition, the consolidated platform’s expanded capabilities can reportedly support more advanced algorithms and features. All software interfaces for new St. Jude Medical pacemakers, ICDs and CRT devices will be the same.
The Promote RF CRT-D reportedly allows physicians to electronically reconfigure left ventricular (LV) leads to help optimize the pacing performance of the device without the need to physically reposition the lead.
In addition, the devices feature the VIP (Ventricular Intrinsic Preference) algorithm that is designed to allow the patient’s own heart rhythm to prevail when possible. VIP technology actively monitors the heart on a beat-by-beat basis to provide pacing only when needed, which has been shown to be better for patients’ overall heart health.


 January 13, 2026
January 13, 2026