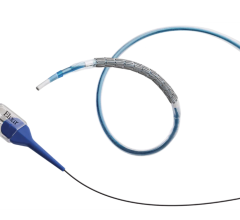
February 5, 2008 – CV Therapeutics Inc. and Medlogics Device Corp. entered into an agreement under which Medlogics has licensed CV Therapeutics’ proprietary biopolymer stent coating technology to develop a drug eluting stent (DES), as Medlogics recently received CE Mark for its Cobalt Super Alloy (COBRA) stent.
CV Therapeutics received Medlogics stock and is entitled to development milestone payments, royalties and other potential payments on future sales of any products incorporating the technology.
Medlogics recently received a CE Mark from European regulatory authorities for its COBRA stent and expects to launch the stent in Europe in the first quarter of 2008. Medlogics expects to utilize CV Therapeutics’ biopolymer stent coating technology to develop a next-generation DES based on the COBRA stent platform.
Based on preclinical data, CV Therapeutics’ proprietary biopolymer stent coating technology reportedly controls the drug release rate more precisely. The proprietary manufacturing process used to create the biopolymer may limit or reduce cracking and peeling following implantation of the stent.
“The novel architecture and thin strut design of our COBRA stent platform, combined with CV Therapeutics’ unique bioerodible technology, provide Medlogics with an outstanding opportunity to develop innovative improved products to meet the needs of interventional cardiologists and their patients,” said Richard L. Klein, president of Medlogics.
For more information: www.medlogics.com and www.cvt.com


 July 02, 2024
July 02, 2024