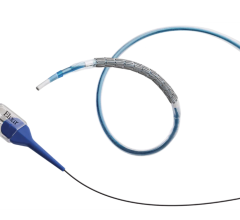
January 23, 2009 - Isotechnika Inc. said yesterday that partner Atrium Medical Corp. has completed patient enrollment for their CONFIRM 1, First-in-Man (FIM) drug-coated coronary stent clinical trial evaluating Atrium's CINATRA voclosporin-coated coronary stent system.
The prospective, multi-center, single blind, randomized, controlled study will compare the CINATRA voclosporin-coated coronary stent system with Atrium's CINATRA bare metal coronary stent (BMS) platform. This first use study of voclosporin on an implantable medical device enrolled 100 patients and is currently being conducted at seven hospitals in Belgium with Glenn Van Langenhove, M.D., PhD of Middelheim Hospital, Antwerp, as the study's principal investigator. The primary endpoint of the trial is to evaluate the safety and performance of the CINATRA voclosporin-coated coronary stent system in patients with de novo coronary artery disease. Patient outcomes, in addition to drug coated stent performance, will be assessed by measuring late lumen loss, in-stent and in-segment lumen loss measurements at six months following implantation.
The CINATRA Voclosporin-Coated Coronary Stent System is comprised of a cobalt chromium bare metal stent mounted on a delivery balloon catheter. The stent and the balloon are coated with Atrium's proprietary Omega-3 fatty acid coating technology utilizing voclosporin for its anti-inflammatory properties.
For more information: www.isotechnika.com, www.atriummed.com


 July 02, 2024
July 02, 2024