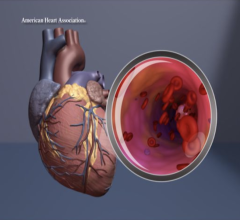
The Corvia intra-atrial shunt to treat diastolic heart failure.
November 25, 2017 – Results presented at the 2017 American Heart Association (AHA) Scientific Sessions and published in Circulation show that a new device designed to treat diastolic heart failure is safe and effective. The first patient in the randomized, blinded study was enrolled at The Ohio State University Ross Heart Hospital, which also enrolled the most patients in the trial.
Diastolic heart failure (DHF) occurs when the heart muscle becomes stiff and does not relax enough to allow blood to flow from the lungs into the heart. This causes blood to back up in the lower left chamber, then the upper left chamber and into the lungs, causing shortness of breath.
“The therapies we use for people with systolic heart failure don’t work as well for people with DHF. For decades, we’ve only been able to manage the symptoms of DHF with diuretics,” said Rami Kahwash, M.D., assistant professor of medicine, director of Ohio State Heart and Vascular Research Organization at Ohio State Ross Heart Hospital.
Kahwash and Scott Lilly, M.D., Ph.D., interventional cardiologist and director of the Structural Heart Disease Program at Ohio State, were part of the Phase II study that evaluated a dime-size implant intended to relieve the high pressure created by blood backing up from the heart into the lungs. During a heart catheterization, doctors inserted the tiny, interatrial shunt device in an opening they created between the upper left and right chambers. This diverts some of the blood from the high pressure left atrial chamber to the low pressure right atrial chamber, much like a detour around a traffic jam.
The study randomized 44 DHF patients at multiple centers nationwide. Participants received either the treatment device or a sham procedure. After one month, patients were evaluated by comparing pulmonary capillary wedge pressure (PCWP) readings during right heart catheterization exercise. Elevated PCWP readings indicate failure of the left ventricle.
Researchers reported that the interatrial shunt device reduced PCWP readings by 3.5 mmHg, compared to a reduction of 0.5 mmHg in the control group.
“This device represents a novel therapy for diastolic heart failure, a condition characterized by lifestyle-limiting breathlessness, and one in which traditional heart failure therapies have not proven effective. If the initial experience is substantiated through ongoing clinical trials, this interatrial shunt device may be an opportunity to improve quality of life and exercise capacity for patients that currently have few other options,” Lilly said.
Risk factors for DHF include high blood pressure, diabetes, obesity and age. Kahwash said it’s estimated that within the next five years, DHF will account for more than half of all heart failure cases, and be responsible for more hospital admissions than systolic heart failure.
Ohio State Ross Heart Hospital is already recruiting patients in the next phase of the clinical trials testing the interatrial shunt device, which is made by Corvia Medical Inc.
Links to other AHA 2017 Late-breaking Trials
Related articles on Heart Failure Device Interventions:
Ohio State Investigating High-Tech Vest for At-Home Heart Failure Management
Ohio State Evaluates First Transcatheter Diastolic Heart Failure Implant
VIDEO "Technologies to Reduce Heart Failure Readmissions," an interview with William Abraham, M.D.
VIDEO "Remote Heart Failure Monitoring Results in Reduced Readmissions" — Abraham discusses the results of the CHAMPION trial of the CardioMEMS system, another heart failure patient monitoring system.


 November 14, 2025
November 14, 2025