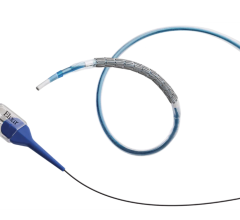
January 3, 2013 — Abbott announced that the Xience Xpedition everolimus-eluting coronary stent system received U.S. Food and Drug Administration (FDA) approval and is launching immediately in the United States, providing physicians with a next-generation technology with the largest size matrix in the U.S. market. Xience Xpedition features a new stent delivery system designed to optimize deliverability, particularly in challenging coronary anatomies.
Samin K. Sharma, M.D., director of clinical and interventional cardiology, dean of international clinical affiliations, and president of the Mount Sinai Heart Network at Mount Sinai Medical Center, New York, was the first physician to implant a patient with Xience Xpedition in the United States.
"Xience Xpedition represents a powerful combination of deliverability, strong clinical data and broad availability of meaningful sizes, three factors that make this product well-suited to treat a wide range of patients," Sharma said. "The impact of the changes to the stent delivery system is particularly notable in patients with complex coronary anatomy. For these patients, physicians have a new technology to reach the blockage and restore blood flow with ease and confidence."
Abbott's Xience drug-eluting stents, including the newly approved Xience Xpedition, are the first and only drug-eluting stents in the U.S. market to be proven safe for direct stenting. Direct stenting, a technique in which the stent system is not preceded by another device (such as a balloon dilatation catheter) to prepare the lesion, has the potential to save time and resources in the catheterization laboratory. Xience Xpedition is supported by rclinical evidence from the Xience family of drug-eluting stents, including data from more than 45,000 patients across more than 100 studies, with long-term outcomes out to five years. Data have consistently shown an excellent safety profile for the Xience family of stents.
"The launch of Xience Xpedition in the United States will advance Abbott's worldwide market-leading position in drug-eluting stents," said John M. Capek, Ph.D., executive vice president, Medical Devices, Abbott. "Xience Xpedition leverages the strong clinical outcomes of Xience V and Xience Prime while providing important advantages in deliverability. With its redesigned stent delivery system and a full matrix of sizes, Xience Xpedition provides physicians with a comprehensive, trusted option to treat a broad range of patients with coronary artery disease."
Xience Xpedition will be available in the largest size matrix in the U.S. market, with both rapid exchange (RX) and over-the-wire (OTW) configurations, providing physicians with the most popular delivery platforms. The stent has diameters ranging from 2.25 mm to 4 mm, including a unique 3.25 mm diameter, and lengths from 8 mm to 38 mm, for more accurate vessel sizing.
For more information: www.abbott.com



 July 02, 2024
July 02, 2024