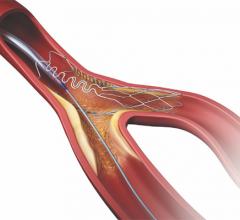
March 10, 2010 - The third annual Workstation Face-Off at the 22nd European Congress of Radiology, held in Vienna, Austria, and hosted by radiologists Anno Graser M.D., and Christoph Becker, M.D., of the Ludwig-Maximillians University in Munich, featured two challenging cases involving oncology and perfusion imaging and analysis with CT and PET, for three sequential timepoints.
As cutting-edge technologies collide with innovative surgical and interventional treatments for multiple coronary or vascular issues, a hybrid approach is emerging as the best path of action.

The general public usually does not take a big interest in cardiology or the interventional devices used to treat cardiac conditions until a family member or someone famous makes headlines due to a cardiac condition. Stents were brought back to mainstream national and international headlines recently after former President Bill Clinton received two stents at New York Presbyterian Hospital.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...

March 9, 2010 – The most recent data from 30 late-breaking clinical trials is being highlighted during the American College of Cardiology (ACC) 2010 scientific sessions, March 14-16 in Atlanta.
March 9, 2010 – As an alternative to the technetium (Tc-99M) isotope used in many nuclear imaging studies, UltraSPECT is advocating the use of thallium isotope (TI-201) imaging as an alternative.
Advanced imaging features are offered by Sonosite for cardiac imaging. These include SonoHD imaging technology and SonoMB multibeam imaging technologies, which provide superior contrast resolution and tissue differentiation, supporting complete diagnostic examinations and ultrasound-guided imaging procedures. The systems also offer customized presets for individual clinical applications.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
March 8, 2010 – Cardiovascular death is the most common cause of mortality among type 2 diabetics and claims the lives of millions each year, with many diabetics experiencing their first “symptom” as a heart attack or sudden death. Understanding that diabetics are at high risk for coronary heart disease (CHD) and many are asymptomatic, Toshiba America Medical Systems Inc.
March 9, 2010 – During the American College of Cardiology (ACC) 59th Annual Scientific Session, March 14-16, CardiacAssist will host its “Clinical Innovations in TandemHeart Support Symposium and User Group Meeting” from 6-8 p.m. Sunday, March 14, at the W Hotel Midtown, Altitude Room, 188 14th St. NE, in Atlanta.
March 9, 2010 — An analysis of six tests that are used to measure platelet function and help gauge the effectiveness of antiplatelet drugs for patients undergoing a cardiac procedure such as a coronary stent implantation found that only three of the tests were associated with a modest ability to predict outcomes such as heart attack or death, according to a study in the Feb.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
March 9, 2010 — Initial clinical study results published in the journal Circulation indicates use of a physician-directed, patient self-management system guided by left atrial pressure can improve symptoms and outcomes in patients with heart failure (HF).
March 2, 2010 — Leg arteries that become narrow decrease blood circulation and can cause these extremities to ache or cramp when a person begins exercise. This condition is referred to as intermittent claudication (IC). Although the pain may subside with rest, IC should be taken seriously because it can increase the risk of heart attack and stroke.
March 8, 2010 – A self-expanding and disconnectable stent today gained CE mark approval in Europe to treat acute coronary syndrome (ACS).
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
March 8, 2010 – Advanced visualization and decision support solutions maker TeraRecon was awarded Frost and Sullivan’s “Company of the Year Award,” in the category of medical imaging, advanced visualization applications, Europe 2010. This award represents the fifth time TeraRecon has been recognized with an award from the healthcare market-consulting firm.
March 8, 2010 – Six-month follow-up data on the Stentys drug-eluting and bare metal stents showed a 4 percent restenosis rate in complex lesions.
March 8, 2010 – Bifurcation stent developer Tryton Medical Inc. recently announced its Tryton stent implants reached a milestone of 750 patients and recently had its first implants in Portugal and Austria.


 March 09, 2010
March 09, 2010