The U.S. Food and Drug Administration (FDA) granted 510(k) clearance to Medical Imaging Electronics (MiE) and Nuclear Imaging Services (NIS) ECAT Scintron. It is the only upgrade path available to the Siemens ECAT 47, ECAT HR+, and ECAT Accel positron emission tomography (PET) systems. The ACSII and SUN workstation are removed from the system and replaced with new PC-based technology that provides new and existing users of the ECAT series PET systems access to faster processing speeds, new acquisition protocols and parts availability. With improved reliability and longevity, the ECAT Scintron redefines the life cycle of dedicated PET systems in the U.S. market.
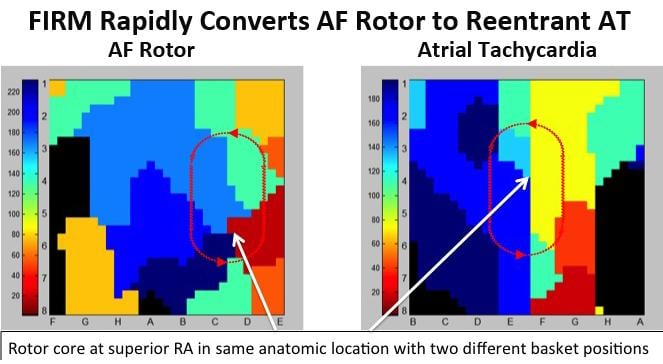
New research presented at Heart Rhythm 2013 continues to show promising results for focal impulse and rotor modulation (FIRM) mapping to effectively target atrial fibrillation (AF) sources and improves ablation therapy outcomes. The novel diagnostic real-time mapping system helps target ablation therapy to patient-specific drivers of AF rather than to anatomical targets, which can improve patient outcomes.
Leviticus Cardio performed a successful animal surgical trial using its wireless coplanar energy transfer system (CET) for ventricular assist devices (VAD). The surgery was performed at Assaf Harofeh Hospital and produced a 75 percent full system efficiency rate as compared with 78 percent efficiency in vitro.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
The incremental mortality in implantable pacemaker and defibrillator recipients who experience a device infection, compared to patients without device infection, is substantial and persists for at least three years after index hospitalization with infection. These are the key findings of a retrospective cohort study of 200,219 Medicare fee-for-service patients undergoing cardiac device procedures, with and without infection, that were presented today by M. Rizwan Sohail, M.D., a researcher from the Mayo Clinic divisions of infectious diseases and cardiovascular diseases, at Heart Rhythm 2013, the Heart Rhythm Society’s 34th Annual Scientific Sessions.
New data from 100,438 patients with Boston Scientific implantable cardioverter defibrillators (ICDs) and cardiac resynchronization therapy defibrillators (CRT-Ds) followed in the Latitude Patient Management System demonstrate the battery life of Boston Scientific single-chamber ICDs, dual-chamber ICDs and CRT-Ds are projected to last an average of 13.2, 11.5 and 9.2 years, respectively

St. Jude Medical gained CE mark approval of its Ilumien Optis percutaneous coronary intervention (PCI) optimization system to better visualize stent planning and navigation. The system integrates both fractional flow reserve (FFR) technology to measure pressure inside the coronary arteries and intravascular optical coherence tomography (OCT) imaging technology, in one system.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
Positron Corp. announced the release of the PosiRx 3000-Series, its latest pharmacy automation systems. The PosiRx 3000-Series are the first systems to automate and encompass the complete compounding process, from generator elution to dose distribution, of multiple diagnostic SPECT agents in an environment engineered to be ISO Class 5 and USP 797 compliant. Designed for facilities dispensing as many as 300 patient-specific doses per day, the PosiRx 3000-Series will benefit providers and patients by enabling unit dose radiopharmaceuticals to be prepared more cost effectively and accurately than previously possible.

Elixir Medical Corp. announced it received CE (Conformité Européenne) mark approval for its DESolve Novolimus-eluting bioresorbable coronary stent scaffold system. The scaffold is designed to degrade in about one year returning the patients’ coronary vessel ultimately to its normal de novo state.
Merck, known as MSD outside the United States and Canada, announced that the U.S. Food and Drug Administration (FDA) has approved Liptruzet (ezetimibe and atorvastatin) tablets for the treatment of elevated low-density lipoprotein (LDL) cholesterol in patients with primary or mixed hyperlipidemia as adjunctive therapy to diet when diet alone is not enough. Liptruzet (pronounced LIP-true-zett) contains ezetimibe, an efficacious LDL cholesterol lowering therapy, and atorvastatin, currently one of the most widely prescribed statins in the United States. Once-daily Liptruzet treats two sources of cholesterol by inhibiting both the absorption of cholesterol in the digestive tract — through ezetimibe — and the production of cholesterol in the liver — through atorvastatin.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...

University of Michigan researchers said the preliminary results of using biresorbable zinc stents were amazing, with corrosion rates exactly where they need to be for a stent successful platform.
Boston Scientific Corporation completed a first-in-human clinical trial utilizing the IntellaTip MiFi XP Ablation Catheter for the treatment of type 1 atrial flutter, an arrhythmia originating in the right atrium of the heart that affects nearly 1 million people in the United States. This single center feasibility trial enrolled 10 patients and was led by Prash Sanders, MBBS, Ph.D., FHRS, director of the Centre for Heart Rhythm Disorders at the University of Adelaide and the Royal Adelaide Hospital in Australia.
Sorin Group has received U.S. Food and Drug Administration (FDA) approval of its Investigational Device Exemption (IDE) application and clinical trial protocol to begin evaluating its Perceval S sutureless aortic tissue valve.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
St. Jude Medical Inc. has received CE Mark approval of its next-generation Ellipse and SJM Assura portfolio of implantable cardioverter defibrillators (ICDs) and cardiac resynchronization therapy defibrillators (CRT-Ds). The new portfolio is designed to lower the risk for lead abrasion and to ensure high-voltage therapy delivery.
Vascular Solutions Inc. announced that it has re-launched the Venture catheter, a deflectable-tip catheter used to provide guidewire directional control in challenging coronary and peripheral interventional procedures. The device is available immediately in the United States and expected to become available in select international markets over the next few months.
The Spectranetics Corporation announced the U.S. Food and Drug Administration's (FDA) approval of its adjunct analysis plan associated with the EXCITE ISR clinical trial, studying the treatment of in-stent restenosis (ISR) in the legs. The goal of the adjunct analysis plan is to demonstrate statistically significant clinical superiority of laser atherectomy plus balloon angioplasty (PTA) compared with PTA alone, prior to full enrollment of the EXCITE ISR study.


 May 17, 2013
May 17, 2013








