The Geneva Healthcare Suite has been proven effective by clinicians at the Douglas & Nancy Barnhart Cancer Center at Sharp Chula Vista Medical Center in managing risk for patients with cardiac devices undergoing radiation therapy.
Healthline today launched its HealthData Engine to harness the power of structured and unstructured data to improve outcomes and reduce costs. The new big-data analytics platform leverages the company’s HealthTaxonomy advanced clinical natural language processing (NLP) technologies and semantic analysis to turn patient data into actionable insights.

In partnership with Intel-GE Care Innovations, Cigna-HealthSpring is expanding its population health program that utilizes interactive tablets – the Intel-GE Care Innovations Guide – and virtual connection to Cigna-HealthSpring nurse practitioners to partner and engage with patients diagnosed with congestive heart failure (CHF) to successfully manage their condition at home. Based on a successful pilot in Middle Tennessee that engaged 50 patients, the innovative remote patient management program is now being implemented with 250 patients statewide with potential for further expansion.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
Researchers have discovered a previously unknown cardiac molecule that could provide a key to treating and preventing heart failure. The newly discovered molecule provides the heart with a tool to block a protein that orchestrates genetic disruptions when the heart is subjected to stress, such as high blood pressure.
Although the highly saturated picture archiving and communication systems (PACS) segment is slowing growth in the U.S. imaging informatics market, a few niche areas are advancing forming new growth areas.
The U.S. Food and Drug Administration (FDA) approved Lumason (sulfur hexafluoride lipid microsphere) for patients whose ultrasound image of the heart (echocardiograms) are hard to see with ultrasound waves.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
Infraredx Inc. announced that its dual-modality intravascular imaging technology, TVC Imaging System, and its TVC Insight Catheter received Shonin approval from the Japanese Ministry of Health, Labour and Welfare (MHLW).
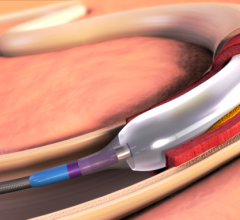
Presented for the first time at the 2014 Transcatheter Cardiovascular Therapeutics (TCT) conference, the latest clinical and economic data on the IN.PACT Admiral drug-coated balloon (DCB) from Medtronic Inc. augments an already robust body of evidence that continues to drive a reconsideration of the standard of care for peripheral artery disease (PAD) in leg arteries above the knee.
Covidien announced that it has notified customers of a voluntary Field Safety Alert for certain Medi-Trace Cadence and Kendall Multi-function Defibrillation Electrodes due to a connector compatibility issue with Philips FR3 and FRx defibrillators.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
October 13, 2014 — After reviewing updated data and analysis for the Boston Scientific Watchman left atrial appendage (LAA) closure device, the U.S. Food and Drug Administration (FDA) Circulatory System Devices Panel of the Medical Devices Advisory Committee voted in favor of the device. By a vote of 6 to 5 (with 1 abstention) the panel concluded the benefits of the Watchman device outweigh the potential risks.
Developed for practices and clinics that do not have a PACS or other disaster recovery solution, Informity uses cloud-based technology to automatically and continuously back up patient information, images and system settings on the ImagePilot Sigma and ImagePilot Aero. It assures business continuance with a one button restore of the entire system.
A report released by the Department of Health and Human Services projects that hospitals will save $5.7 billion this year in uncompensated care costs because of the Affordable Care Act, with states that have expanded Medicaid seeing about 74 percent of the total savings nationally compared to states that have not expanded Medicaid.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
A study using a new imaging tool, optical coherence tomography (OCT), offered a new look at the composition of carotid artery disease and has the potential to alter how physicians understand and treat the disease, a leading cause of stroke.
Although cardiovascular disease is largely avoidable through lifestyle modifications, it remains the nation’s number one cause of death. While annual wellness exams offer physicians the chance to advise patients on modifying cardiac risk factors, that advice can easily get lost given the amount of information covered during a routine check-up.
“Once patients have survived cancer, they don't die from cancer, they die from heart disease. Cardio-oncology is about making sure that doesn't happen," said Juan Carlos Plana, M.D.

 October 15, 2014
October 15, 2014