DrChrono Inc. and 3D4Medical have teamed up so practices across the United States can access 3-D interactive modeling and animation videos from within their electronic health record (EHR) to better educate patients.

The choices we make every day can have a lasting effect on our heart and vascular health. Adopting a heart healthy eating plan, getting more exercise, avoiding tobacco and managing known risk factors are among the key recommendations in the 2019 Primary Prevention of Cardiovascular Disease guideline from the American College of Cardiology (ACC) and the American Heart Association (AHA). Also, it is recommended that aspirin should only rarely be used to help prevent heart attacks and stroke in people without known cardiovascular disease.

As many as one in four patients who undergo cath lab interventions can benefit from a technology that identifies the ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
Researchers from the Stanford University School of Medicine presented preliminary results of the Apple Heart Study, a virtual study with over 400,000 enrolled participants, at the American College of Cardiology (ACC) 68th Annual Scientific Sessions, March 16-18 in New Orleans. The researchers reported that wearable technology can safely identify heart rate irregularities that subsequent testing confirmed to be atrial fibrillation, a leading cause of stroke and hospitalization in the United States.

Virtual reality (VR) and its less immersive kin, augmented reality (AR), are gaining traction in some medical ...

Machine learning is already having an enormous impact on cardiology, automatically calculating measurements in ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
March 15, 2019 – Biotronik announced U.S. Food and Drug Administration (FDA) approval of the Acticor and Rivacor high ...
The American College of Cardiology’s first annual ACC Quality Summit, held March 13-15 in New Orleans, merges the NCDR (National Cardiovascular Data Registry) Annual Conference and ACC Accreditation Congress for the first time to optimize cardiovascular patient care. The conference brings together cardiovascular thought leaders, administrators, clinicians, clinical quality leaders and data quality experts.
March 15, 2019 — Artificial intelligence (AI) company Bay Labs announced the presentation of two studies assessing ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
March 14, 2019 — Beginning in mid-2019, hospitals performing transcatheter valve repair and replacement will be able to ...
Siemens Healthineers will introduce the Somatom go.Top Cardiovascular Edition, a new version of its established computed tomography (CT) system, at the 68th Annual Scientific Session and Expo of the American College of Cardiology (ACC), March 16-18 in New Orleans. The Somatom Go.Top Cardiovascular Edition is designed to deliver personalized patient dose control in all types of routine cardiovascular imaging. The cost-effective, 128-slice scanner offers outpatient cardiology offices as well as hospitals access to not just coronary CT angiography (CCTA), but also advanced tests, including the HeartFlow FFRCT (fractional flow reserve computed tomography) Analysis.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
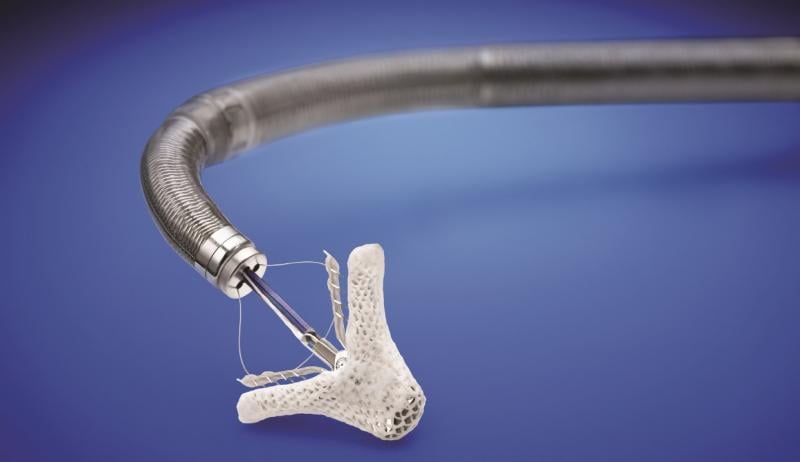
March 14, 2019 — The U.S. Food and Drug Administration (FDA) approved the MitraClip heart valve repair device for ...
An imaging procedure commonly performed before starting cancer treatment can provide valuable clues about a patient's risk for heart problems in the months and years after treatment. However, this information is not always reported and is rarely acted upon in current practice, according to research being presented at the American College of Cardiology's 68th Annual Scientific Session, March 16-18 in New Orleans.
V-Wave Ltd. announced that renowned heart failure cardiologist William T. Abraham, M.D., is joining V-Wave as chief medical officer. Abraham makes this move to V-Wave after more than 16 years as director of the Division of Cardiovascular Medicine at the Ohio State University.


 March 18, 2019
March 18, 2019











