Medtronic's Defender embolic protection filter, available in Europe, is a device designed to get rid of emboli that can occur during a variety of stenting procedures.
April 29, 2008 - HealthEast Care System publicized The National Brain Aneurysm Center and the www.brainaneurysmcenter.org Web site, formerly the HealthEast Neurovascular Institute, focusing on complex neurovascular conditions such as aneurysm, stroke, vascular malformations, brain tumors and more.
April 29, 2008 - The FDA has approved the LipiScan Coronary Imaging System, an angiography laser scanner by InfraReDx Inc., which is intended to characterize fatty deposits in coronary vessel walls using near infrared diffuse spectroscopy.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
April 28, 2008 - Minimally invasive heart robotic bypass surgery could lead to faster recoveries for patients and a ...
Armstrong Medical will display its Auto-Locking Cart with an optional proximity card reader for quick access at the 2008 ...
April 28, 2008 - Terumo Heart will present data on its DuraHeart LVAS, a circulatory support device intended to provide ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
April 28, 2008 - Researchers at the University of Warwick have found a that the protein E-selectin is a marker for endothelial dysfunction and sub-clinical systemic inflammation, which can help identify a far greater number of people at high risk for future development of type 2 diabetes.
April 28, 2008 - Medtronic Inc. today said the FDA has cleared the OneTouch UltraLink Meter as the new wireless meter ...
Medtronic Inc. has expanded its product line to include the OneTouch UltraLink Meter as the new wireless meter ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
April 28, 2008 – A study of clinical trials involving artificial blood substitutes has shown they can cause an increased ...
April 28, 2008 - There was no difference in mortality between men and women for patients who suffered a myocardial infarction, according to a study in Germany that included 132,000 men and women.
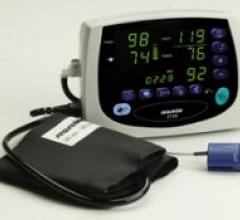
The multi-function Avant 2120 tabletop pulse oximeter combines Nonin Medical’s PureSAT pulse oximetry technology with ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
April 28, 2008 - Doctors at The National Brain Aneurysm Center in St. Paul, MN, use intraoperative angiography (IA) now ...
April 25, 2008 - EPIX Pharmaceuticals Inc. met all pre-specified endpoints prospectively agreed to with the FDA in a ...
April 25, 2008 - CV Therapeutics reported the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMEA) recommended approval of ranolazine in the European Union (EU) for the treatment of chronic angina.

 April 28, 2008
April 28, 2008