The push for healthcare to be digitally transformed and paper-free means having access to electronic medical records anytime/anywhere. This effort amplified, has left many cardiology departments struggling to catch up. According to the Administration on Aging people 65+ represented 12.4 percent of the population in the year 2000, but are expected to grow to be 19 percent of the population by 2030. The increase in longevity-on top of advances in medications, less invasive treatments and diagnostic testing will greatly increase demand for cardiology.
Blackford Analysis, a provider of software products that accelerate comparison of medical images, introduced its products to the Middle Eastern market at the Arab Health Congress 2014 in Dubai. Designed to be integrated directly into any image viewer, such as a PACS, Advanced Visualization Viewer or Universal Viewer, Blackford Analysis’ products work within existing systems to enable instant comparison of multiple image studies.
The U.S. Food and Drug Administration (FDA) Medtronic Inc.’s Valiant Captivia Thoracic Stent Graft System to treat type B aortic dissections. Supported by the results of the U.S. Medtronic DISSECTION trial, the new indication provides physicians a minimally invasive alternative to open surgical repair and medical therapy.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...

Radiation dose continues to rise as the number of computed tomography (CT), nuclear, angiography and fluoroscopy examinations grow, leading to a greater risk of patient overexposure to radiation. Healthcare providers must reinforce their efforts to monitor and visualize dose levels from radiology examinations to enhance patient safety and meet new regulatory demands. There also is a need to justify and optimize the usage of radiation dose to find a balance between safer practice, image quality and lower dose — all for the benefit of the patient. Implementing tools for automatic and continuous follow up of radiation dose is at the forefront of meeting these challenges.
Instead of making a chest incision for open heart surgery, cardiovascular surgeons can now use a new vacuum-like tool to remove potentially deadly, large clots that lodge in patients’ hearts.
A large-scale analysis assessing the real-world risk of venous thromboembolism (VTE) in chemotherapy patients shows a greater occurrence of VTE than identified in clinical trials. Risk progressively increases during the year following treatment initiation, according to the report.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
Physicians have used radiation in medicine for more than a century. The use of radiation in diagnostic imaging, including computed tomography (CT), fluoroscopy, angiography, mammography, computed radiography (CR) and digital radiography (DR), as well as in nuclear medicine, has aided greatly in the diagnosis and treatment of cancer and other diseases.
Ekos Corp., a BTG International group company, published results of its Ultrasound Accelerated Thrombolysis of Pulmonary Embolism (PE) trial (ULTIMA) in the peer-reviewed journal of the American Heart Association, Circulation. The announcement was made at the International Symposium on Endovascular Therapy (ISET), Miami, Fla.
Miracor Medical Systems completed four months’ follow-up of patients enrolled in the “Prepare RAMSES” clinical trial of PICSO (Pressure-controlled Intermittent Coronary Sinus Occlusion) System.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Heart transplant patients may live 20 years or more after surgery, according to a study in The Annals of Thoracic Surgery. Hector Rodriguez Cetina Biefer, M.D. and Markus J. Wilhelm, M.D., from the University Hospital Zurich in Switzerland, led a research team that examined long-term outcomes in 133 patients from their institution who underwent heart transplantation from 1985 to 1991.
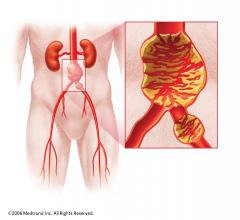
W. L. Gore & Associates Inc. (Gore) and AAAneurysm Outreach have partnered to raise awareness and drive screening of individuals at-risk of abdominal aortic aneurysm (AAA).
Ascendian Healthcare Consulting announced a service line to assist clients with planning, design, development and implementation of Cardiovascular Information Systems (CVIS).
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...

Expanded use of percutaneous coronary intervention (PCI) is seeing more patients with acute coronary syndromes treated more quickly, according to the latest National Audit of PCI (covering 2012).

New leaders, new categories, new Best in KLAS segments and an aethetic overhaul are some of the highlights of the 2013 Best in KLAS Awards: Software & Services report.
The Centers for Medicare and Medicaid Services (CMS) released findings on a number of its initiatives to reform the healthcare delivery system. These include interim financial results for select Medicare accountable care organization (ACO) initiatives, an in-depth savings analysis for Pioneer ACOs, results from the physician group practice demonstration, and expanded participation in the bundled payments for care improvement initiative. Savings from both the Medicare ACOs and Pioneer ACOs exceed $380 million.

 February 03, 2014
February 03, 2014