CardioDx Inc. is a molecular diagnostics company specializing in cardiovascular genomics. It published an analysis demonstrating that the Corus CAD gene expression score is significantly associated with plaque burden and luminal stenosis. The holds true in symptomatic patients being evaluated for obstructive coronary artery disease (CAD), as measured by CT-angiography. The study, "A Peripheral Blood Gene Expression Score is Associated with Atherosclerotic Plaque Burden and Stenosis by Cardiovascular CT-Angiography," appears online in the journal Atherosclerosis.
Biotronik, a manufacturer of implantable cardiac devices and wireless remote monitoring technologies, has been added to the list of cardiac rhythm management (CRM) device suppliers to the U.S. Department of Veterans Affairs (VA).

Advancements in technologies are paving the way for many healthcare providers to connect with patients outside their hospitals more quickly and efficiently. For cardiology departments, technologies that allow for video collaboration, rapid transmission of scans and images and other forms of telemedicine are supporting the initiative to improve productivity, an initiative many are striving for as the U.S. healthcare landscape continues reform.
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
At Johnston Medical Center in Smithfield N.C., StatVideo’s EchoCart streams real-time images of babies’ hearts and live video conferencing over the web to pediatric cardiologists at Duke Children’s Hospital. EchoCart is a telemedicine system designed specifically for tele-echocardiography.
Vascular Solutions Inc. launched the ThrombiDisc topical hemostat designed for use around indwelling lines up to 12°F. ThrombiDisc contains both the power of thrombin to facilitate hemostasis and the antimicrobial properties of silver.
After several months of intense review and assessment of its specialized worker training program by an independent agency, Operon Resource Management of Lowell, Mass. received ISO 13485 certification.
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
A University of Utah-led study for treatment of patients with atrial fibrillation (A-fib) provides strong clinical evidence for the use of 3-D MRI to individualize disease management and improve outcomes. Results of the Delayed-Enhancement MRI Determinant of Successful Radio-frequency Catheter Ablation of Atrial Fibrillation (DECAFF) study will be published in the Journal of the American Medical Association.
eCardio Diagnostics is opening its second Independent Diagnostic Testing Facility (IDTF) in San Francisco, Calif. Its first IDTF is located in Houston, Texas.
Results from several late breaking clinical trials will be presented during the American College of Cardiology (ACC) 2014 annual meeting March 29-31. These will be featured during five late-breaking clinical trial sessions.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
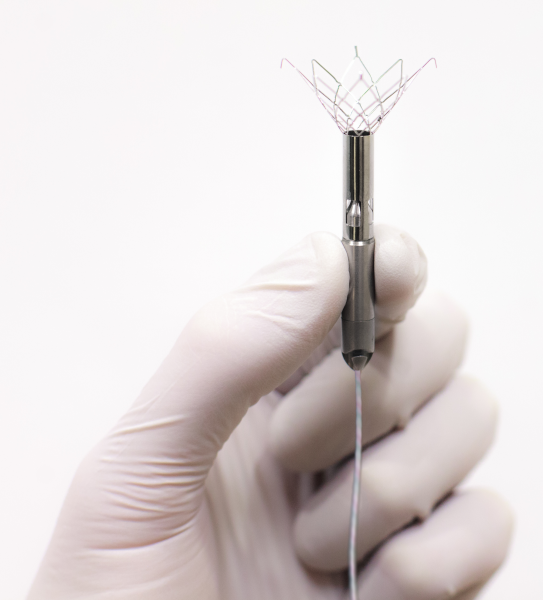
Start-up company Procyrion Inc. is developing a catheter-deployed circulatory assist device intended for long-term use in the treatment of chronic heart failure. The 6 mm diameter Aortix device is narrower than a pencil and is delivered via a catheter in a minimally invasive outpatient procedure lasting about 10 minutes.
The American College of Radiology (ACR) strongly supports the bicameral, bipartisan legislation to replace the sustainable growth rate (SGR) payment formula. The organization particularly applauds inclusion of several ACR backed provisions that raise quality of care, make care more efficient and increase transparency in physician payment policy.

Doctors at Henry Ford Hospital in Detroit used an Edwards Lifesciences Sapien transcatheter aortic valve replacement (TAVR) device to repair a mitral valve that was narrowed with calcium buildup. William O'Neill, M.D., medical director of the Center for Structural Heart Disease at Henry Ford Hospital, estimates this new technique could help thousands of patients a year in the United States.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
Transcatheter Technologies GmbH, a medical device company, is developing a third-generation transcatheter aortic valve implantation (TAVI) system, Trinity. An independent laboratory completed advanced wear testing (AWT) of the company’s Trinity valve prosthesis. AWT of the Trinity heart valve has completed 600 million cycles, or an estimated 15 years of durability testing.

The topic of radiation safety and radiation dose monitoring has moved from state-specific regulations to a national trend with The Joint Commission’s (TJCs) recent announcement of their “New and Revised Diagnostic Imaging Standards.” The call for dose management and tracking has graduated from being advised to being mandated – from both a legal perspective and from within the world of healthcare’s patient safety foundation. The question that many organizations find themselves asking is “Where does this leave me?” and “Are we prepared for compliance?”
To address the needs of physicians who treat patients with valvular heart disease, Esaote North America established a sales force to bring 3mensio Structural Heart software for cardiovascular planning solutions.

 February 07, 2014
February 07, 2014