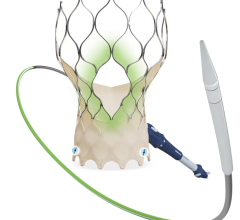
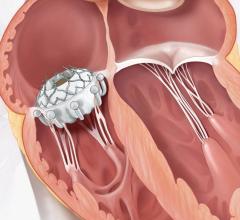
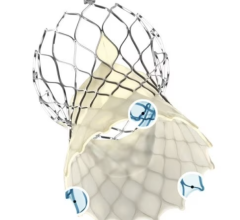
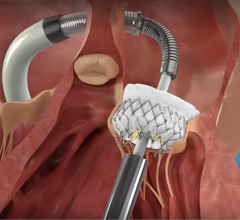
May 6, 2024 — Teleflex Incorporate, a leading global provider of medical technologies, today announced that the Wattson Temporary Pacing Guidewire limited market release has commenced at Columbia University Irving Medical Center. The first cases were performed by Dr. Tamim Nazif, Director of Clinical Research, Dr. Susheel Kodali, Professor of Medicine and Director of the Structural Heart and Valve Center, and Dr. Isaac George, Surgical Director of the Heart Valve Center.
Featuring a simple design to create procedural efficiencies, the Wattson Temporary Pacing Guidewire offers dual functionality, supporting both valve delivery and ventricular bipolar pacing during structural heart procedures, including transcatheter aortic valve replacement (TAVR) and balloon aortic valvuloplasty (BAV). The device offers a procedural alternative designed to avoid potential complications1, steps1, and costs2 associated with traditional right ventricular pacing. From its flexible distal pigtail shape to its multiple electrode, bipolar design, the Wattson™ Temporary Pacing Guidewire is engineered to help reduce the risk of ventricular perforation while providing confidence in capture during rapid pacing.
“We are thrilled that our team at Columbia University Irving Medical Center was selected for the Wattson Temporary Pacing Guidewire limited market release and are proud to have completed the first TAVR procedures with this important new device,” said Dr. Tamim Nazif, Director of Clinical Research for Columbia Interventional Cardiovascular Care, Director of the Interventional Cardiology Fellowship Program, and assistant professor in medicine at Columbia University Irving Medical Center. “The Wattson Temporary Pacing Guidewire facilitated minimalist TAVR procedures with safe and reliable LV pacing throughout our first two days of cases, and we are eager to continue to use it moving forward.”
Teleflex received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the Wattson Temporary Pacing Guidewire in May of 2023.
“Providing interventionalists with a new tool specifically engineered to address unmet clinical needs frequently encountered during structural heart procedures is a significant milestone for Teleflex,” said Roger Graham, President and General Manager of the Teleflex Interventional business unit. “The Wattson Temporary Pacing Guidewire reflects our steadfast focus on filling gaps in existing technologies as well as our commitment to providing more options that further simplify minimalist TAVR and other structural procedures.”
For more information: www.teleflex.com
References:
- Clinical Investigation: TEMPORARY PACING GUIDEWIRE STUDY. Testing completed by Teleflex. Data on file with Teleflex. Bench test results may not necessarily be indicative of clinical performance.
- Faurie B, Souteyrand G, Staat P, Godin M, Caussin C, Van Belle E, Mangin L, Meyer P, Dumonteil N, Abdellaoui M, Monségu J, Durand-Zaleski I, Lefèvre T; EASY TAVI Investigators. Left Ventricular Rapid Pacing Via the Valve Delivery Guidewire in Transcatheter Aortic Valve Replacement. JACC Cardiovasc Interv. 2019 Dec 23;12(24):2449-2459.


 July 08, 2024
July 08, 2024